Overall Objectives
This clinical study was initiated as part of the national effort to respond
the coronavirus (COVID-19) pandemic due to the SARS-CoV2 virus.
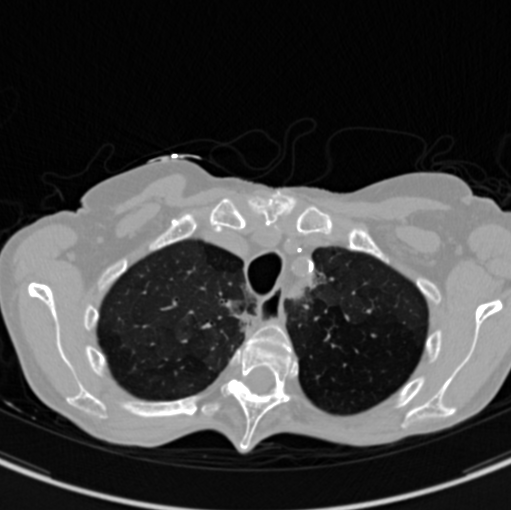
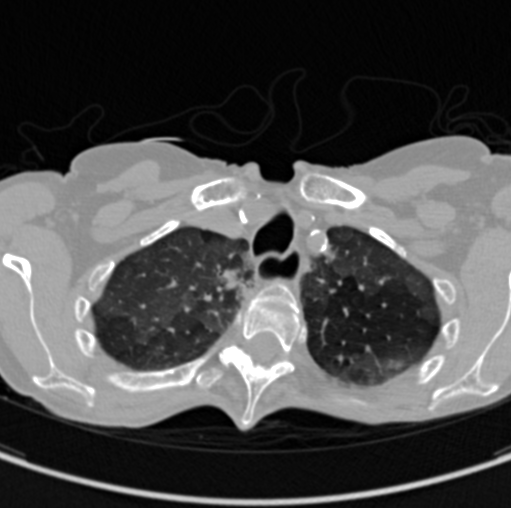
COVID-CTPRED is a comprehensive cohort database gathering an enhanced and
comprehensive chest-CT imaging together with biological,patient history, risks factors, and current clinical data.
The overall objectives is to refine our understanding, identify composite prognostic factors of
the disease, and help triage with “all-round” patients, with non-COVID viral infiltrates and
pneumopathies.
Computational medical imaging alone has shown its ability to predict a therapeutic response
or a particular evolution after extracting from the volume of images informations that are
not visually perceptible during standard radiological reading, making it possible to construct
a radiomic signature and/or using anatomical/functional metrics.
By combining these data extracted from the scanner with the standard clinical-biological
data produced at admission during triage, our ambition is to build more representative
predictive models to anticipate clinical evolution and to optimize the management of the
resources
The University Hospital of Saint-Etienne sponsors this study
(ClinicalTrials.gov Identifier NCT04377685).
This plateform
-
- Is the entry point to the entire database containing all pseudonymized chest-CT images and corresponding clinical report form (CRF) data.
- In order to guarantee patients' rights with regard to the promoter's commitments to the protection of health data, and in particular because of their exhaustive nature, CREATIS has set up a service making available a dedicated internalized computing architecture for launching external algorithms and deep learning models